 
Of particular interest, the three new drugs were substantially less likely than warfarin to cause a particular kind of bleeding leading to stroke – a “hemorrhagic stroke,” a stroke caused by bleeding into the brain, which is different from the strokes caused by the clots that go to the brain in atrial fibrillation. On the basis of clinical trials that included more than 50,000 patients from around the globe, FDA concluded that all three drugs were either equivalent to, or more effective than, warfarin in preventing strokes, with an acceptable risk of bleeding. Like warfarin, all three are ‘blood thinners’ that reduce the overall risk of stroke related to atrial fibrillation but they also cause bleeding. In the last few years, FDA has approved three new oral anticoagulant drugs – Pradaxa (dabigatran), Xarelto (rivaroxaban), and Eliquis (apixaban). Thus, the dosage of warfarin must be carefully adjusted to keep the blood thinning effect in the right range. 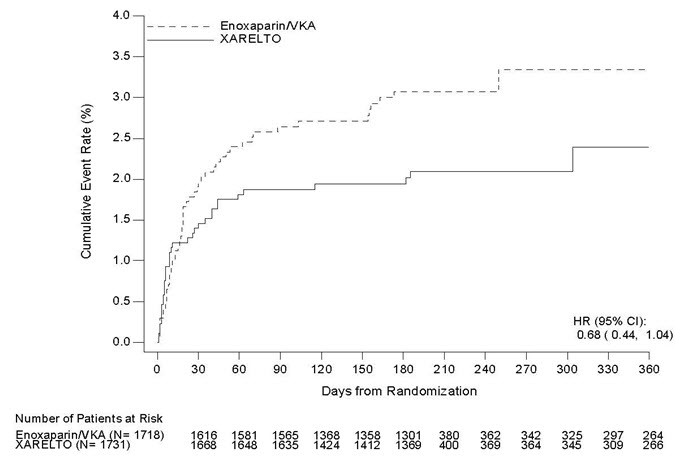
If the effect is too small, it will fail to prevent strokes if the effect is too high, it will cause excess bleeding. But the anticoagulant effect of warfarin must be carefully monitored with periodic blood tests. Until recently, warfarin (approved in 1954 and marketed under the brand names Coumadin and Jantoven) had been the only drug approved for the prevention of stroke in patients with atrial fibrillation. This is accomplished with taking anticoagulant drugs or ‘blood thinners.’ Decreasing stroke riskĪnticoagulants have been known for many years to produce a striking (more than 50%) decrease in the rate of stroke, but they also prevent clotting in locations and situations where clotting is desirable. There are a number of treatments - drugs and procedures - intended to correct the fundamental heart rhythm problem in patients with atrial fibrillation, but the main focus of treatment is to try to decrease the rate of stroke by preventing the formation of these blood clots. The main concern, however, is that atrial fibrillation can lead to the formation of blood clots in the heart, which can travel to the brain and cause a stroke. 
Atrial fibrillation can produce palpitations, shortness of breath, lightheadedness, weakness, and chest pain, or may occur without symptoms. They are accused of downplaying the risk of bleeding to promote Xarelto as superior to warfarin, a drug that is easily de-activated with a dose of Vitamin K.More than 3 million Americans have atrial fibrillation, a problem with the electrical system of the heart that causes an irregular heart rhythm. The manufacturers of Xarelto, Bayer and Janssen Pharmaceuticals, are facing about 8,000 lawsuits involving severe injuries and deaths from uncontrollable bleeding. In the European Union alone, an estimated 73,000 patients on these drugs will be hospitalized for major bleeding or require urgent surgery. Without a way to de-activate their blood-thinning medication, doctors may struggle to stop bleeding or delay a life-saving surgery. Portola said 1-4% of patients on Factor Xa inhibitors experience major bleeding, and another 1% need emergency surgery. Because none of them have reversal agents, AndexXa is an FDA-designated “breakthrough” drug. The company sought approval for AndexXa after two Phase III clinical trials showed that it reversed Eliquis by 93% and Xarelto by 92% for two hours in healthy patients.įactor Xa inhibitors are a new class of blood-thinning drugs that have only been on the market since 2011. The FDA asked Portola for more information on manufacturing and data to support the inclusion of Savaysa and Lovenox on the label. The surprise decision was announced yesterday by Portola Pharmaceuticals. AugThe FDA has delayed approval for AndexXa (andexanet alfa), the first-ever antidote to stop uncontrollable bleeding in patients on Xarelto and Eliquis. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
